Direct steric hindrance and steric acceleration both have been found with suitably placed large substituents and reagents, and indirect effects arising because one group interferes with the orienting power of another also are known. Steric (spatial) effects generally are smaller than electronic effects in determining the characteristics of reaction by this mechanism, but they are not negligible. These effects are very specific with regard to the position at which the modifying group is introduced. As would be expected, substituents that release electrons toward the reaction site facilitate the reaction, and those that withdraw electrons retard reaction. Reactivity by this mechanism is dominated by the electrophilic character of the reagent (E) however, it also responds powerfully to changes in structure of the organic substrate. 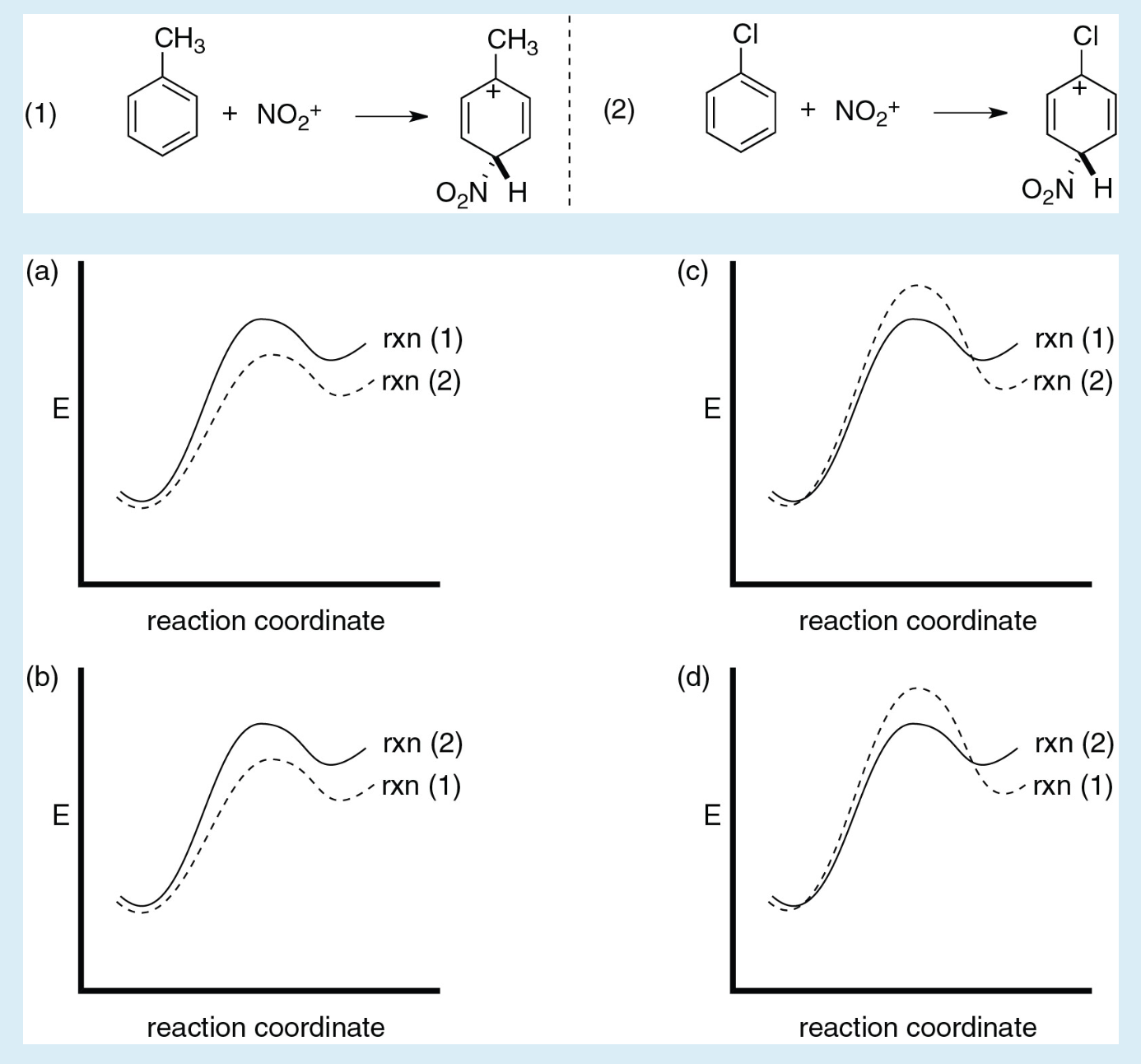
Examples are known in which the removal of the proton from the carbonium ion intermediate (to form the second pi complex) becomes rate-determining. This complex forms rapidly in an equilibrium preceding the rate-determining step, which itself leads to a carbonium ion intermediate and then by way of a second pi complex to the product. Here, Y represents a substituent on the ring the arrow from the ring centre indicates coordination.Īs shown, the reaction begins with formation of a pi complex, in which the electrons associated with the aromatic ring, or other unsaturated centres (pi electrons), coordinate weakly with the electrophile. Both substituents can be any one of various groups (e.g., hydrogen atoms or nitro, bromo, or tert-alkyl groups). One of the more common is shown here reactions in this category consist of replacement of a group designated Y (often a hydrogen atom) in an aromatic molecule by an electrophilic agent designated E. Reaction by any one of several mechanisms is possible. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!Įlectrophilic substitution at unsaturated carbon centresīecause of its wide applicability, particularly to aromatic systems, electrophilic substitution is an important reaction.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Britannica Beyond We’ve created a new place where questions are at the center of learning.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find.Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives.Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions.This one and the last one will be the primary. This will be followed by 1 c: 6 h, 5 ch, 3 b r. Only the 3 degree 1 that is given is this 1 c, 6 h, 5 c, with c h, 3 with c 6 h, 5b r. In the 1 case there are 2 fenyle rings, so that will be more stable. We can see that the stability order is 3 degree, 2 degree and one degree. On the other side, we can see that the last compound is attached to hydrogen on one side and to ch 3 on the other. This side is attached to bromine and the final group side. It is attached to the final group on the other side. The carbon atom is attached to a piece of wood. On the other side, it is attached to c 6 h, 5 and the side we are on now. The second one that has given carbon is attached to the other one. The first compound that is given is c: 6 h, 5 ch, and 2 b r. 3 degree followed by 2 degree and 1 degree is all. Let's write the stability order of carbocation because the more reactive will be the compound. The more stable the cargo cet ion is, the more we need to arrange them according to the reactivity order of the reaction. As different compounds are given different, different compounds are given, and what we are supposed to do is arrange them in their reactivity order of the s n 1 reaction, what all is given in the problem.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
